Peptide radioreceptor therapy (PRRT) with Lutetium-177 DOTATOC
The radioactive medication Lutetium-177 DOTATOC is used for this purpose. This substance is very similar to the hormone somatostatin and binds to the receptors on the surface of tumor cells. Coupling the radionuclide to this substance (peptide) enables therapeutically effective radiation doses to act directly on the tumor in a very specific way and with the best possible protection for the healthy tissue.
Preparation for a therapy Lu-177
In order to be able to assess whether PRRT with Lu-177 DOTATOC is possible and indicated in each individual case, a detailed anamnesis of the previous course of the disease is required.
Several examinations are required to plan and prepare for therapy:
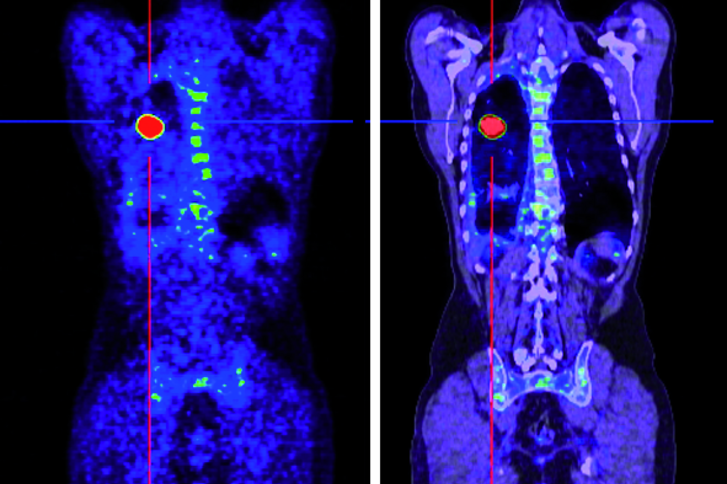
- somatostatin-receptor-scintigraphy and a gallium-68 DOTATOC PET / CT examination to document sufficient storage in the tumor lesions
- Blood tests: blood count, liver and kidney values, tumor markers
- Up-to-date kidney scintigraphy (Tc-99m MAG-3 and Tc-99m DTPA) to evaluate the kidney function
The therapy cannot be performed in one of the following cases:
- insufficient storage of somatostatin analogues in the tumor lesions
- severe renal insufficiency
- urinary stasis kidneys
- uncontrollable urinary incontinence
- serious changes in the blood count
Risks and side effects of Lu-177
The therapy is generally very well tolerated, but the following side effects can occur:
- nausea, vomiting, diarrhea, fever
- liver dysfunction
- renal dysfunction up to renal insufficiency (loss of function of the kidney), which we counteract with a kidney protecting infusion during therapy
- allergic reactions up to anaphylactic shock (this is very unlikely, countermeasures can be taken immediately)
- changes in the blood count, in extreme cases bone marrow damage and degradation of all blood cells, malignant neoplasms cannot be completely excluded even many years after the therapy course.
The inpatient stay in Helios Berlin-Buch
The patient will be admitted to our therapy ward for 3 days.
Our ward at Helios Berlin Buch consists of 11 rooms with shower, toilet, television and telephone. Due to the radiation protection rules, relatives are not permitted to visit this ward. As patients have to be in the room and on the ward, they are kindly asked to bring comfortable clothing. The patient is also welcome to bring personal belongings like books etc. The patient can use your mobile phone and computer in the room as well as in the patient lounge. There is free WiFi access on the ward.
The personal belongings can usually be taken back home after the inpatient stay.
Implementation of Lu-177 therapy
For intravenous therapy it is necessary for the ward doctor to place an indwelling venous cannula in patient's arm. The therapeutic substance is slowly administered intravenously as an infusion using an infusion pump over a period of 30 minutes. Since the radioactive therapeutic substance is excreted via the kidneys, the patient will receive an amino acid infusion before, during and after the therapeutic infusion in order to protect the kidneys from possible damage.
After the therapy, various nuclear medicine recordings (full-body scintigraphy / SPECT / CT) are taken about 30 minutes later to document and control the activity distribution and to measure the radiation exposure of the kidneys. In order to document the course of therapy, recordings are also made on the gamma camera after 24, 48 and 72 hours.
If our patients have any questions, please do not hesitate to contact our ward via telephone 030-9401- 53460.
After therapy
After the treatment, blood values should be checked after 2 weeks, in particular blood count, liver and kidney values, in order to identify any side effects that may occur and to be able to react to them.
If peptide radioreceptor therapy (PRRT) with Lutetium-177 DOTATOC is well tolerated, 4-6 treatment cycles with an interval of 3 months are generally planned. In order to be able to evaluate the therapy response, and in preparation for the following therapy, it is necessary to carry out the above-mentioned preliminary examinations repeatedly before each cycle.
Legal aspects
The therapeutic substance is not a commercial preparation of the pharmaceutical industry. The therapeutic agent is produced individually for you in the radiopharmacy of the Clinic for Nuclear Medicine of the Helios Clinic Berlin-Buch. By producing it on site, your attending doctor assumes individual responsibility for the for the preparation and use of the medicinal product (Section 13.2b of the Medicines Act).
The treatment performed as part of a so-called individual healing attempt.
Read more:
Do you need more information about Helios Hospitals or want to schedule your treatment?